
AstraZeneca’s tozorakimab meets COPD trial endpoints
📚 Related People & Topics

AstraZeneca
British-Swedish pharmaceutical company
AstraZeneca plc () (AZ) is a Swedish-British multinational pharmaceutical and biotechnology company with its headquarters at the Cambridge Biomedical Campus in Cambridge, UK. It has a portfolio of products for major diseases in areas including oncology, cardiovascular, gastrointestinal, infection, ...
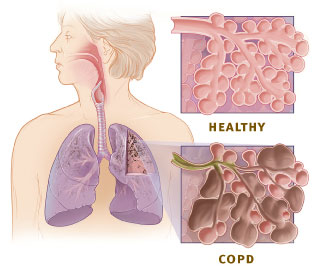
Chronic obstructive pulmonary disease
Lung disease involving long-term poor airflow
Chronic obstructive pulmonary disease (COPD) is a long-term lung condition that makes it progressively harder to breathe. It occurs when the airways and lung tissue become damaged, leading to reduced airflow and less efficient oxygen exchange in the lungs. COPD develops gradually over time, and whi...
Entity Intersection Graph
Connections for AstraZeneca:
Mentioned Entities
Deep Analysis
Why It Matters
This development matters because chronic obstructive pulmonary disease (COPD) affects over 300 million people globally and is the third leading cause of death worldwide. Tozorakimab's success in clinical trials represents a potential breakthrough treatment option for patients with limited therapeutic alternatives, particularly those with severe forms of the disease. The positive results could significantly impact AstraZeneca's respiratory medicine portfolio and competitive position in the pharmaceutical market. This news affects COPD patients, healthcare providers, investors, and competing pharmaceutical companies developing similar therapies.
Context & Background
- COPD is a progressive lung disease characterized by breathing difficulties, chronic cough, and mucus production, often caused by long-term exposure to irritants like cigarette smoke
- Current COPD treatments primarily focus on symptom management through bronchodilators and corticosteroids, with limited disease-modifying options available
- AstraZeneca has been expanding its respiratory portfolio through both internal development and acquisitions, competing with companies like GSK and Novartis in this therapeutic area
- Tozorakimab is a monoclonal antibody targeting IL-33, a key inflammatory pathway implicated in several respiratory diseases including COPD and asthma
- The global COPD treatment market was valued at approximately $15 billion in 2023 and is projected to grow significantly due to aging populations and continued smoking prevalence in some regions
What Happens Next
AstraZeneca will likely proceed with regulatory submissions to health authorities including the FDA and EMA in the coming months, potentially leading to market approval within 12-18 months if the data supports it. The company may initiate additional phase 3 trials for other respiratory indications like severe asthma. Competitors developing similar IL-33 inhibitors will need to reassess their development strategies based on these results. Healthcare providers will await detailed data publication to understand which patient subgroups benefit most from this therapy.
Frequently Asked Questions
Tozorakimab is a monoclonal antibody developed by AstraZeneca that targets interleukin-33 (IL-33), a protein that plays a key role in inflammatory responses in respiratory diseases. By blocking IL-33, the drug aims to reduce airway inflammation and potentially modify disease progression in COPD rather than just treating symptoms.
These results are significant because they demonstrate efficacy in a disease area with limited new treatment options. If approved, tozorakimab would represent one of the first biologic therapies specifically targeting the underlying inflammatory mechanisms of COPD, potentially offering a new approach for patients who don't respond adequately to existing treatments.
Key challenges include demonstrating long-term safety and cost-effectiveness compared to existing therapies, identifying the specific patient populations that benefit most, and navigating regulatory requirements for a new mechanism of action in COPD. The drug will also face competition from other emerging biologics and established inhaled therapies.
This strengthens AstraZeneca's respiratory portfolio alongside existing products like Symbicort and Fasenra. Success with tozorakimab could establish AstraZeneca as a leader in biologic treatments for respiratory diseases and provide a significant revenue stream as patents expire on some of their older respiratory medications.
Based on typical biologic development patterns, tozorakimab will likely target patients with moderate-to-severe COPD who experience frequent exacerbations despite standard therapy. The drug may be particularly beneficial for patients with specific inflammatory profiles or those who have elevated biomarkers related to IL-33 pathway activation.